By Anthony House
See chart - (Fig. 1 Liquors - from spagyric lesson 41).
You can see on this chart provided by LPN France that, distilled vinegar of red wine; alkahest of tartar; alkahest of niter; alkahest of sulfur; alkahest vinegar of antimony; philosophical mercury from glass of antimony; philosophical mercury of tin; and philosophical mercury of mercury, are virtually universal in scope in the vegetable, mineral, and metallic kingdoms.
Each having its advantages and limitations.
The exceptional liquor for universal coverage (extraction of metallic oils) being the philosophical mercury of tin.
PURPOSE
I want these metallic oils for further research in medicinal and initiatory aims.
Tiny samples of the raw ores may be extracted in test tubes for far reaching study.
Initially, in 1991 I wanted to make the alkahest of native mineral sulfur, since it’s supposed to extract the oils of the seven planetary metals, except gold; but including antimony.
I used alcohol at 96% and sealed some ground crystals of native sulfur [the crystals of sulfur should be separated from the limestone completely] in a honey jar. I left them to work in a warm area of my kitchen cabinet.
This was started on 3-11-91. I periodically checked for the formation of sulfur of sulfur crystals to form on the surface of the philosophical alcohol, but only found a tiny amount formed there, even after nearly three years time.
So I opened the jar and transferred the contents into a roomy 1 liter retort.
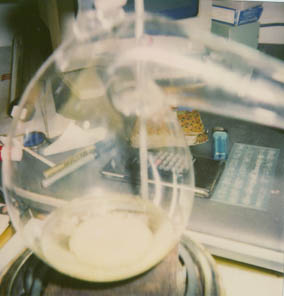
I loosely fitted a 1 liter receiving flask (round boiling flask) in a water bath, to the neck of the retort. (See Fig. 2).
The neck didn’t have a ground glass joint, and I didn’t want any overpressure to cause an explosion (the alkahest has a hell of a kick when it manifests!).
Mineral lesson #3 tells us to do this work in a closed system, but since this was my first attempt I thought it prudent to first distill the alcohol off at a very low temperature (just enough for vapors to rise), and disregarded the atmospheric contamination of water in the alcohol. It was also felt that the experiment would yield a way of obtaining more nascent crystals of native mineral sulfur.
I found that crystals formed more rapidly than in the closed jar I had used previously, (the lesson states that it can take as long as two years for crystals to form).
Crystals also formed on the glass of the retort as well, since saturation was happening.
After two complete lunar cycles starting at the new moon, the 10th of February 1994, I noticed that the alcohol had indeed absorbed some water.
The water contained crystals that floated on the surface, and oil was also floating around in it.
In order to retrieve the oil/ alcohol, and water mix, I re-imbibed the distilled alcohol on the crystals and again distilled until most of the alcohol had gone over and the water and oil was apparent, one last time.
Using a pipette, I drew the water and oil out of the retort through the opening in the top of the retort and put the mixture in a bottle that had a ground glass stopper for storage.
Now I had some red drops (globules) of sulfur oil. (Some floats on the surface and most sinks to the bottom). Note that in general, oils will float in water and oil sinks in alcohol. If oils sink in water, it means that they’re heavy with salts.
QUERY
Is this the formation of sulfuric acid mentioned in the lessons? Or is it oil of native sulfur? The appearance of the oil is very organic, belying the fact that it came out of a mineral.
It’s asserted that this alkahest, even in tiny amounts, can extract native sulfur swiftly.
Further tests will have to be conducted. I ostensibly obtained a small amount of alkahest, I believe, that was present in the alcohol.
Although, this part of the procedure doesn’t give the completed alkahest.
I venture that some alkahest was obtained, because a few cc’s of the alcohol was poured into a flask that was previously frosted by crystals that couldn’t be removed by any reagent I tried on it.
Yet, about 2 cc’s of the mentioned alcohol immediately and completely removed the crystalline frost from the glass. The crystals didn’t dissolve but were removed whole.
CONCENTRATING THE TINCTURE
I was unhappy with the small amount of sulfur of sulfur crystals formed, so I placed whole native sulfur crystals with 40 ml of fresh 96% alcohol and 10 ml of the former alcohol in a 1 liter boiling flask.
I set a liebig condenser above the flask as a refluxing apparatus. No water was circulated to cool the condenser though.
I used a stopper on the condenser to close it off (to keep the atmospheric moisture out).
More than once the stopper went flying, and left a dent in the ceiling above the refluxer where the stopper had hit!
When it popped out of the end of the condenser a thick smoke would form in the boiling flask as air rushed in. This is a sign that alkahest is evident.
I kept the temperature very low to keep the pressure from building up, but, the alkahest was undoubtably forming in small volume.
At this stage in the study, I found that little outside heat was needed to be applied to the flask.
Perhaps because the alcohol is so pure and the alkahest is so very volatile, that a concentration of the tincture can be had without even distillation heat.
CONCLUSION
This concentration method proved to be an excellent way to condense the sulfur of sulfur crystals by adding enough sulfur (native sulfur crystals crushed or uncrushed) into the alcohol to form a dense tincture.
The more alcohol that’s added results in dissolving the crystals that have already formed.
I also tried evaporating some tincture in an open dish, but, all of the tincture was vaporized, leaving no crystals.
I’ll be starting some experiments with the apparatus shown in the lesson, - with the tiny crystals, and more formed crystals soon, making sure to set up a shield in front of the closed device for protection!
A later report will be presented in this forum for study.
SOURCES: LPN Spagyric lesson 41; LPN Mineral lesson 3.





This page has been visited times.